Original Article
Optic Nerve Diseases and its
Systemic Associations
Mubashir Rehman, Akhundzada Muhammad Aftab, Sher Akbar Khan, Imran
Ahmad
Pak J Ophthalmol 2017, Vol. 33, No. 3
. . . . . . . . . . . . . . . . . . . . . . .
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .. .
.. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
|
See end of article for
authors affiliations
…..………………………..
Correspondence to:
Mubashir Rehman
Eye
Department
LGH/PGMI, Lahore
E.mail: drmubashirrehman78@gmail.com
|
Purpose: To determine
optic nerve diseases and its systemic associations.
Study Design: Descriptive cross sectional
study.
Place and Duration of Study: Eye
departments of Lady Reading Hospital Peshawar and Khyber Teaching Hospital
Peshawar from Jan 2015 to Oct 2015.
Material and Methods: A total of 44 patients were
examined. Detailed history was taken from every patient after which complete
ocular examination including recording of visual acuity, checking pupillary
reactions and fundoscopy with special attention to optic nerve head
appearance and recording of color vision and light brightness sensitivity was
carried out. Specific ophthalmic and systemic investigations were performed.
Results: Seven (15.91%) patients had
NAION, 6 (13.64%) had demyelinating optic neuritis, 2 (4.54%) had toxic optic
neuropathies, 2(4.54%) had nutritional optic neuropathy, 3 (6.82%) had
pituitary macroadenoma and 3 (6.82%) had benign intracranial hypertension, 2
(4.54%) had arteritic anterior ischemic optic neuropathy, 2 (4.54%)
Pseudo-Foster Kennedy syndrome, 1 (2.27%) had paraneoplastic syndrome, 1
(2.27%) had superior saggital sinus thrombosis, 1 (2.27%) had occipital lobe
infarct, 1 (2.27%) had brain metastasis, 1 (2.27%) had craniopharyngioma, 1
(2.27%) had bilateral thalamic ischemia and1 (2.27%) had hydrocephalus.
Conclusion: Patients
presenting with optic nerve diseases may have serious
systemic associations so for accurate diagnosis and management every patient
presenting with optic nerve disease must be properly evaluated.
Key
words: Optic nerve, optic neuritris, optic neuropathy.
|
One of the frequent causes of visual
loss encountered by ophthalmologists is optic neuropathy1. Clinically, it can appear as an isolated entity due to local
pathologies of the optic nerve or associated to a variety of
systemic illnesses2.
Optic neuropathy may be unilateral or bilateral and usually
presents with swelling of the optic disc or atrophic optic disc. However it is
not uncommon for an optic nerve disease that optic nerve head appear clinically
normal but it may cause other signs of optic nerve damage such as decreased
visual acuity and defective color vision or light brightness sensitivity or
presence of relative afferent papillary defect3.
Optic neuropathy has a number of local and underlying systemic
causes including ischaemia, demyelinating disease, multiple sclerosis, systemic
lupus erythematosus (SLE), sarcoidosis, Behçet’s disease, vasculitis, and
several infections including lyme disease, syphilis and cat scratch fever2.
Recognition of the underlying cause can not only change the visual
prognosis but also the neurological consequences. Thus ophthalmologist should
therefore be familiar not only with the various entities that can cause optic
neuropathy but also should have knowledge of relative systemic investigations
to diagnose a systemic illness which may be the cause for optic nerve damage4. In
most cases, careful clinical evaluation and appropriate investigations, can
lead to a specific diagnosis5.
Similarly as the optic nerve dysfunction may be the presenting
sign of a systemic illness, knowledge of clinical evaluation of the optic nerve
and appropriate and relative investigations is also important for physicians
because physicians may be the first person to encounter such patient and if
misdiagnosed this entity can end up in blindness and optic atrophy apart from
systemic sequel4.
Purpose
of our study was to find out various local as well as underlying systemic
causes giving rise to optic nerve damage and to emphasize the significance of
early diagnosis and hence timely management with the help of clinical signs and
relative ophthalmic as well as systemic investigations which can not only
prevent blindness but also help diagnosis and management of underlying systemic
disease.
MATERIAL
AND METHODS
It was a descriptive cross sectional study carried out at
department of ophthalmology of Lady Reading Hospital Peshawar and Khyber
Teaching Hospital Peshawar from Jan 2015 to October 2015. A total of 44
patients including males and females were examined. Sample size was
calculated using 95%
Confidence interval and 10% margin of error, under WHO sample size estimation. All patients presenting with sudden or gradual loss of vision
with optic nerve involvement evident by decrease vision, presence of affarant
pupillary defect, defective color vision, reduced light brightness sensitivity
and contrast sensitivity and optic nerve appearance, were included in the
study. Patients with all other causes of decreased vision with or without
presence of affarant papillary defect e.g. diabetic and hypertensive
retinopathy, central retinal vein occlusion, central retinal artery occlusion
and retinal detachment were excluded from the study. Detailed history was taken from every patient
after which complete ocular examination including recording of visual acuity,
pupillary reactions, intraocular pressure, recording of color vision and light
brightness sensitivity and fundus examination with special attention to optic
nerve head appearance was carried out for every patient. Specific ophthalmic
investigations like automated visual field analysis (Humphrey) and Optical
Cohenrence Tomography (OCT) (Optic Nerve Protocol) were carried out where
needed. Systemic investigations in
collaboration with physicians were performed for individual cases based upon
their history, ophthalmic and systemic examination.
All the
analysis was done in SPSS version 20.0. For categorical variables like gender,
and local and systemic associations, frequencies and percentages were
calculated. For numeric variables like age, mean ± standard deviation was
calculated. All the results were presented in the form of tables.
RESULTS
A total
of 44 patients were included in this study. Age distribution is shown in table
1. Mean age was 44.09 years with SD ± 17.47. Gender distribution was analyzed
as n = 24 (54.54%) of patients were males and n = 20 (45.46%) were females.
Table 1: Age Distribution.
|
Age
|
Frequency
|
Percentage
|
|
10 – 20 Years
|
2
|
4.54%
|
|
21 – 30 Years
|
9
|
20.46%
|
|
31 – 40 Years
|
12
|
27.28%
|
|
41 – 50 Years
|
4
|
9.10%
|
|
51 – 60 Years
|
5
|
11.36%
|
|
61 – 70 Years
|
10
|
22.72%
|
|
71 – 80 Years
|
2
|
4.54%
|
|
Total
|
44
|
100%
|
Mean
age was 44.09 years with SD ± 17.47
Out of 44 patients, 25 (56.81%) patients had local optic nerve
pathology resulting in either optic disc swelling or optic disc atrophy. While
19 (43.19%) patients had underlying systemic illness resulting in either optic
disc swelling, papilledema or optic disc atrophy.
Leading cause of local optic disc pathologies resulting in optic
disc swelling was non arteritic anterior ischemic optic neuropathy (NAION) 7
(15.91%), followed by demyelinating optic neuritis 6 (13.64%). A complete
breakdown of different local causes is given in Table 2.
Local optic disc pathologies resulting
in pale disc included; 2 (4.54%) patients had anterior ischemic
Table 2: Local Optic Disc Pathologies Causing Swollen Disc.
|
Disease
|
Frequency
|
Percentage
|
Presentation
|
|
Non arteritic anterior
ischemic optic neuropathy
|
7
|
15.91%
|
Swollen disc
|
|
Demyelinating optic
neuritis
|
6
|
13.64%
|
Swollen disc
|
|
Optic neuritis
|
2
|
4.54%
|
Swollen disc
|
|
Arteritic anterior
ischemic optic neuropathy
|
2
|
4.54%
|
Swollen disc
|
|
Pseudo-Foster Kennedy
syndrome
|
2
|
4.54%
|
Rt swollen and Lt pale
disc
|
|
Optic nerve drusen
|
1
|
2.27%
|
Swollen disc
|
|
Total
|
20
|
45.46%
|
|
Table 3: Systemic Associations of Bilateral swollen Discs/ Papilloedema.
|
Disease
|
Frequency
|
Percentage
|
Presentation
|
|
Benign intracranial hypertension
|
3
|
6.82%
|
Bilateral swollen discs
(Papilloedema)
|
|
Superior saggital sinus thrombosis
|
1
|
2.27%
|
Bilateral swollen discs
(Papilloedema)
|
|
Brain metastasis
|
1
|
2.27%
|
Bilateral swollen discs
(Papilloedema)
|
|
Known case of chronic renal failure
|
1
|
2.27%
|
Bilateral swollen discs
|
|
Total
|
6
|
13.64%
|
|
Table 4: Systemic Associations of Bilateral pale Discs.
|
Disease
|
Frequency
|
Percentage
|
Presentation
|
|
Pituitary macroadenoma
|
3
|
6.82%
|
Bilateral temporal
disc pallor
|
|
Nutritional optic
neuropathy due to Vit B12 deficiency
|
2
|
4.54%
|
Bilateral temporal
disc pallor
|
|
Nutritional optic
neuropathy due to tobacco amblyopia
|
1
|
2.27%
|
Bilateral temporal
disc pallor
|
|
Occipital lobe infarct
|
1
|
2.27%
|
Bilateral temporal
disc pallor
|
|
Craniopharyngioma
|
1
|
2.27%
|
Bilateral pale discs
|
|
Bilateral thalamic ischemia
|
1
|
2.27%
|
Bilateral pale discs
|
|
Hydrocephalus
|
1
|
2.27%
|
Bilateral pale discs
|
|
Total
|
10
|
22.72%
|
|
optic neuropathy, 2 (4.54%) had traumatic optic neuropathy, while
1 (2.27%) patient with pale disc was undiagnosed.
3 (6.82%) patients with underlying systemic diseases resulted in
swollen disc; 1 (2.27%) patients had toxic optic neuropathy due
anti-tuberculous drugs, 1 (2.27%) had paraneoplastic syndrome secondary to
squamous cell carcinoma of lung and 1 (2.27%) had neurosarcoid.
Leading causes for patients with underlying systemic diseases
resulting in bilateral swollen discs or papilloedema included; 3 (6.82%) benign
intracranial hypertension, followed by superior saggital sinus thrombosis 1
(2.27%), and brain metastasis 1 (2.27%). Complete breakdown of all the systemic
causes has been shown in Table 3.
Patients
with underlying systemic diseases resulting in pale discs included; 3 (6.82%)
had pituitary macroadenoma, 2 (4.54%) had nutritional optic neuropathy due to
vitamin B12 deficiency. Further details of this group are given in table 4.
DISCUSSION
A
variety of intrinsic, intraorbital, intracranial, or systemic diseases can lead
to optic nerve damage1. Misdiagnosis of optic nerve diseases is not
uncommon and can lead to sight as well as life threatening conditions6.
Reduced blood flow to the optic nerve’s ganglion cells can lead to
ischemic
optic neuropathy which may be either non-arteritic (NAION) or arteritic
(AION). NAION may result from a variety of underlying systemic conditions
while AION is caused by giant cell arteritis (GCA)7. Most common
form of ischemic optic neuropathy is NAION. Risk factors include hypertension,
diabetes, hyperlipidemia, atherosclerosis, nocturnal hypotension, sleep apnea,
certain medications and small discs. Behbehani
R in his study on ischemic optic neuropathies commented on the appearance of
the optic disc4. According to him the optic disc is usually swollen
in non-demyelinating optic neuritis and NAION. In addition, in NAION, disc
swelling can be sectoral or diffuse and associated with peripapillary
hemorrhages. A small cup-to-disc ratio is seen in the fellow eye. While
patients with AION due to GCA shows diffuse “chalky white” swelling of the disc
with cotton wool spots4. In addition patients with NAION have
afferent pupillary defect and corresponding visual field loss8.
Although any type of visual field defect can occur with any type of optic
neuropathy, in ischemic optic neuropathies altitudinal visual field defects are
more common9. In our study the criteria we used for diagnosing
non-arteritic anterior ishchemic optic neuropathy (NAION) was relevant clinical
history, decreased visual acuity, presence of relative afferent pupillary
defect, diffuse or sectoral optic nerve head swelling consistent with non
arteritic anterior ischemic optic neuropathy and altitudinal field defect on
Humphery’s visual field. All patients with non arteritic anterior ischemic
optic neuropathy (NAION) in our study were uncontrolled diabetics with
hyperlipidemia. Humphrey’s visual fields showed altitudinal field defect in
four of these patients.
Giant cell arteritis (GCA) should strongly be considered in
patients older than 60 years with features of ischemic optic neuropathy10.
Patients with GCA typically experience headache, scalp tenderness, jaw
claudication, weight loss, fever and malaise. Complete blood count, erythrocyte
sedimentation rate (ESR), and C-reactive protein (CRP) should always be
performed in all such cases. ESR and CRP when combined increases the
specificity (97.0%) and sensitivity (99%) for diagnosis. Thrombocytosis is also
a positive finding in patients with GCA7. Our criteria for
diagnosing arteritic anterior ischemic optic neuropathy was positive clinical
history with special emphasis to jaw claudication, headache, scalp tenderness
and weight loss, reduced visual acuity, presence of afferent pupillary defect,
edematous optic nerve head consistent with arteritic anterior ischemic optic
neuropathy, raised ESR and C- reactive proteins and histological confirmation
of giant cell arteritis on temporal artery biopsy. In our study both patients
with arteritic anterior ischemic optic neuropathy had raised ESR and C-
reactive proteins and temporal artery biopsy revealed calcification.
The only patient of Pseudo-Foster Kennedy syndrome in our study
had optic atrophy on one side and swollen disc on other side with altitudinal
field defect on Humphery’s visual fields on the side of swollen disc with
normal MRI brain and orbits. Optic neuritis refers to an infective, inflammatory, or
demyelinating process affecting the optic nerve. It usually presents in the
second to fourth decades of life11. Most common cause of optic
neuritis is Multiple sclerosis (MS)12. It usually presents with
acute unilateral vision loss, pain on eye movement, presence of afferent
pupillary defect and visual field defects. Optic nerve head may be swollen in
the acute stage but if the optic nerve inflammation is retrobulbar than
appearance of the optic nerve head is unremarkable. Optic nerve head may show
signs of pallor when the acute stage subsides11. Behbehani
R showed in his study that in retrobulbar demyelinating optic neuritis, optic
disc is normal in 65% of cases and even if optic disc is swollen is it is
usually diffuse and of mild degree4. Presence of severe optic disc
swelling with peripapillary hemorrhages and exudates should point to an
alternative diagnosis such as non arteritic AION or infiltrative optic
neuropathy4. Our criteria
for diagnosing demyelinating optic neuritis was positive clinical history,
reduced visual acuity, presence of relative afferent papillary defect, color vision
and light brightness sensitivity defect, visual field defect and positive MRI
findings. In our study all patients with demyelinating optic neuritis had
positive MRI report of high signal intensity lesions in the intra orbital
portion of optic nerves in addition to other positive finding of optic nerve
damage. Two patients in our study with non demyelinating optic neuritis were
diagnosed on clinical basis with reduced visual acuity, color vision and light
brightness sensitivity defect, presence of relative afferent papillary defect
and swollen optic disc with normal investigations.
Optic nerve
head drusen consists of calcific hyaline-like material within the optic nerve
head substance. At an early age these are called as “buried drusen” as these
are not usually visible. At later age these enlarge and come closer to the
surface of optic nerve head and become more visible. B-scan show high acoustic
reflectivity due calcific deposits and is the most reliable method for
diagnosis13. In our study patient with optic nerve drusen presented
with bilateral crowded disc resembling papilloedema but had positive finding on
B-scan of high acoustic reflectivity.
Long-standing optic nerve damage such as caused by nutritional or
toxic optic neuropathies, compressive, traumatic or hereditary optic
neuropathies can give rise to a pale optic disc. This can also occur when an
acute inflammatory or ischemic stage of optic neuropathy subsides14.
Toxic or nutritional and hereditary optic neuropathies selectively affecting
the papillo-macular bundle can give rise to temporal optic disc pallor as
mentioned by Behbehani
R in his study4.
Patients
with traumatic optic neuropathy usually have suffered facial or orbital trauma.
Main clue to the diagnosis is presence of RAPD. To detect fractures of the
optic canal, and to rule out orbital hemorrhage, CT scan orbit is the
investigation of choice15. In our study both the patients with
traumatic optic neuropathy presented to us very late due to other injuries
occurred during road traffic accident. Both had decreased visual acuity,
presence of relative afferent papillary defect and optic disc pallor on the
affected side with normal CT scan brain and orbits. Figure 1 shows fundus
photos of patient with traumatic optic neuropathy.
Different
drugs and nutritional deficiencies can also cause Optic nerve damage. Top on
the list are ethambutol and amiodarone. Tobacco, methanol and ethanol are also
in the list16. Tobacco-alcohol amblyopia is the consequence of toxic
effect of tobacco superimposed on nutritional deficiency state.17
Nutritional optic neuropathy mainly occurs due to vitamin deficiency.
Deficiency of thiamine (vitamin B1), riboflavin (vitamin B2), niacin (vitamin
B3), pyridoxine (vitamin B6), cyanocobalamin (vitamin B12) have all been
implicated16. In our study among patients with toxic and nutritional
optic neuropathies one was heavy smoker with tobacco amblyopia and had
bilateral temporal disc pallor with central scotoma on Humphrey’s visual
fields, one had toxic optic neuropathy resulting from anti-tuberculous drugs
and two had nutritional optic neuropathy due to vitamin B12 deficiency. Our
criteria for diagnosing nutritional optic neuropathies were reduced visual
acuity, temporal or complete disc pallor and serum B12 level below
normal limits. Both our patients with nutritional optic neuropathy due to
vitamin B12 deficiency were low in B12 level with bilateral temporal disc
pallor.
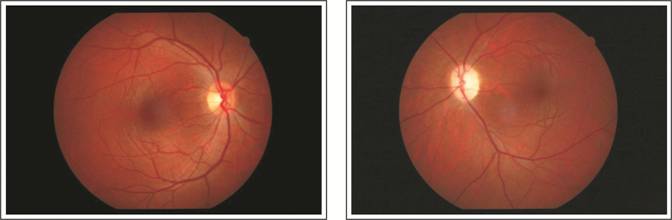
Figure 1: Patient with traumatic
optic neuropathy showing left pale disc.

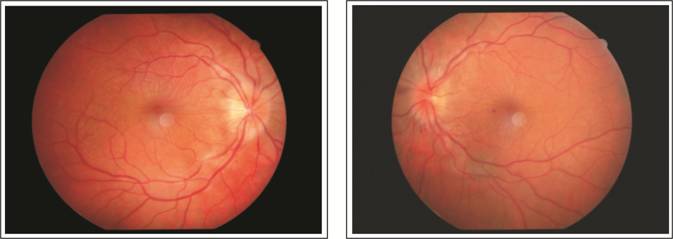
Swollen disc Left lower lobe
consolidation MRI chest
showing growth in left lung
Figure 2: Patient with paraneoplastic
syndrome.
Bilateral swollen discs
(Papilloedema)

MRI showing non visualization
of superior saggital sinus.
Figure 3: Patient with superior saggital
sinus thrombosis.
Both
small cell and non-small cell carcinoma of the lungs can cause optic neuropathy
as a result of paraneoplastic syndrome. Patients usually present with gradual
decrease of vision associated with bilateral disc swelling before the signs of
the systemic malignancy are evident18. In our study patient with
paraneoplastic syndrome presented with left swollen disc. Findings were not
consistent with local optic disc pathologies so we decided to perform systemic
evaluation in collaboration with our physician colleagues. On routine
investigations x-ray chest showed left lower lobe consolidation. MRI chest was
advised which showed growth in left lung for which biopsy was performed which
proved to be squamous cell carcinoma of lung, figure 2.
In our study criteria for diagnosing benign intracranial
hypertension was positive clinical history of headache, presence of
papilloedema, normal MRI with absence of intracranial mass lesion or
enlargement of ventricles, raised opening pressure of CSF on lumber puncture or
clinical trial of acetazolamide with improvement of sign and symptoms in those
patients who were unwilling for lumber puncture. Out of three patients, in our
study, two had raised opening pressure of CSF on lumber puncture while one
patient was advised lumber puncture but was reluctant. Diagnosis in this case
was made on clinical basis and showed improvement with oral acetazolamide.
Three patients presented to us with papilloedema, including
superior saggital sinus thrombosis, brain metastasis and one undiagnosed
patient who had chronic renal failure and investigations were incomplete due to
lack of follow-up. Figure 3 shows papilloedema secondary to superior saggital
sinus thrombosis with positive MRI finding of non visualization of superior
saggital sinus.
Compressive optic neuropathy usually results in gradual and
progressive visual loss19. Common causes include pituitary adenomas,
meningiomas, intracranial aneurysms, craniopharyngiomas and gliomas of the
anterior visual pathway20. Although knowledge of visual field
defects leads in the localization of the lesion, MRI of the brain and orbit is
require for accurate diagnosis19. In our study seven patients presented
with progressive visual loss and bilateral disc pallor which included three
cases of pituitary macroadenoma and one each; occipital lobe infarcts,
craniopharyngioma, bilateral thalamic
ischemia and hydrocephalus. All these cases had positive findings on humphrey’s
visual fields and MRI brain and orbits.
CONCLUSION
Optic nerve diseases may have serious systemic associations and
whether localized or associated with systemic illnesses, it has serious
ophthalmic and systemic sequel so every patient with optic nerve disease must
be properly examined and proper investigations must be performed for accurate
diagnosis and management.
Author’s Affiliation
Dr.
Mubashir Rehman
MBBS,
FCPS, (Ophthalmology) Assistant professor,
Nowshera
Medical College and Hospital, Nowshera.
Dr.
Akhundzada Muhammad Aftab
MBBS, FCPS, (Ophthalmology) Senior registrar,
Khyber Teaching Hospital, Peshawar.
Dr.
Sher Akbar khan
MBBS, FCPS, (Ophthalmology) District specialist, Saidu
Group of Teaching Hospital Sawat.
Dr.
Imran Ahmad
MBBS,
FCPS, (Ophthalmology) Assistant professor, Gaju Khan Medical
College, Swabi
Role of Authors
Dr.
Mubashir Rehman
Patient’s selection, data collection, results & discussion
Dr.
Akhundzada Muhammad Aftab
Patient’s selection, data collection, results & discussion
Dr.
Sher Akbar khan
Patient’s selection, data collection, results & discussion
Dr.
Imran Ahmad
Literature
search
REFRENCES
1.
O'Neill EC,
Danesh-Meyer HV,
Connell PP,
Trounce IA,
Coote MA,
Mackey DA,
Crowston JG. The optic nerve head in acquired optic neuropathies. Nat Rev Neurol. 2010; 6: 221-36.
2.
Cornblath,
Wayne T. Introduction to visual loss.
Neuro-Ophthalmology, 2009; 15: 13-21.
3.
Mackenzie PJ,
Mikelberg FS. Evaluating Optic Nerve Damage: Pearls and Pitfalls. Open
Ophthalmol J. 2009; 3: 54–58.
4.
Behbehani
R. Clinical
approach to optic neuropathies. Clin Ophthalmol. 2007; 1: 233–246.
5.
O'Neill EC,
Danesh-Meyer HV,
Kong GX,
Hewitt AW,
Coote MA,
Mackey DA,
Crowston JG. Optic Nerve Study
Group. Optic disc evaluation in optic neuropathies: the optic disc
assessment project. Ophthalmology, 2011; 118: 964-70.
6.
Wu Y, Zhou LM,
Lou H, Cheng JW,
Wei RL. The Association between Obstructive Sleep Apnea and Nonarteritic
Anterior Ischemic Optic Neuropathy: A Systematic Review and Meta-Analysis. Curr Eye Res.
2016; 41: 987-92.
7.
Hayreh SS,
Zimmerman MB. Optic disc edema in non-arteritic anterior ischemic optic
neuropathy. Graefes Arch Clin
Exp Ophthalmol. 2007; 245: 1107-21.
8.
Sohan Singh Hayreh. Management of ischemic optic neuropathies. Indian J Ophthalmol.
2011; 59: 123–136.
9.
Edward J. Atkins,
Beau B. Bruce, Nancy J. Newman,
Valérie Biousse.
Treatment of Nonarteritic
Anterior Ischemic Optic Neuropathy. Surv Ophthalmol. 2010; 55: 47–63.
10.
BiousseV, Nancy J, Newman. Ischemic Optic Neuropathies. N Engl J Med. 2015; 372: 2428-2436.
11.
Pérez-Cambrodí RJ,
Gómez-Hurtado
Cubillana A, Merino-Suárez ML,
Piñero-Llorens DP,
Laria-Ochaita C. Optic
neuritis in pediatric population: a review in current tendencies of diagnosis
and management. J Optom. 2014; 7: 125-30.
12.
Preziosa P,
Comi G,
Filippi M. Optic neuritis in multiple
sclerosis: Looking from a patient's eyes. Neurology,
2016; 87: 338-9.
13.
Lam BL,
Morais CG Jr,
Pasol J. Drusen of the optic disc. Curr Neurol Neurosci Rep.
2008; 8: 404-8.
14.
Orssaud C,
Roche O,
Dufier JL. Nutritional optic neuropathies. J Neurol Sci. 2007; 262: 158-64.
15. Mackiewicz J,
Tomaszewska J,
Jasielska M. Optic nerve
avulsion after blunt ocular trauma - Case report. Ann Agric Environ
Med. 2016; 23: 382-3.
16.
Sharma
P, Sharma
R. Toxic optic neuropathy. Indian
J Ophthalmol. 2011; 59: 137–141.
17.
Jyoti Prakash,
VSSR Ryali,
K. Srivastava,
P. S. Bhat,
R. Shashikumar,
and A. Singal. Tobacco-alcohol amblyopia: A rare complication of prolonged
alcohol abuse. Ind Psychiatry J. 2011; 20: 66–68.
18.
Ikawa M,
Kuriyama M. Paraneoplastic retinopathy and optic neuropathy. Brain Nerve,
2010; 62: 371-6.
19.
Chiang Ling Koay,
Fiona Lee Min Chew,
Kheng Yaw Chong,
and Visvaraja Subrayan. Compressive optic neuropathy: A unique presentation of Sweet
syndrome. Indian J Ophthalmol. 2013; 61: 140–141.
20.
Cheour M,
Mazlout H,
Agrebi S,
Falfoul Y,
Chakroun I,
Lajmi H,
Kraiem A. Compressive optic neuropathy secondary to a pituitary
macroadenoma. J Fr Ophtalmol. 2013; 36: 101-4.



